The first C2 selective halide substitution reaction of 2,3-epoxy alcohols by the use of (CH3O)3B–MX (X=I, Br, Cl) system.
Tomata, Y.; Sasaki, M.; Tanino, K.; Miyashita, M.
Tetrahedron Lett. 2003, 44, 8975. DOI: 10.1016/j.tetlet.2003.10.001

Total Synthesis of Scytophycin C. 2. Coupling Reaction of the C(1)−C(18) Segment and the C(19)−C(31) Segment, a Key Macrolactonization, and the Crucial Terminal Amidation Reaction.
Nakamura, R.; Tanino, K.; Miyashita, M.
Org. Lett. 2003, 5, 3583. DOI: 10.1021/ol0352299
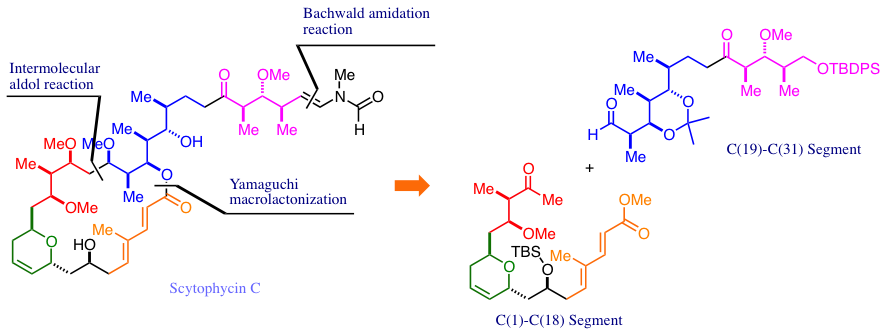
Total Synthesis of Scytophycin C. 1. Stereoselective Syntheses of the C(1)−C(18) Segment and the C(19)−C(31) Segment.
Nakamura, R.; Tanino, K.; Miyashita, M.
Org. Lett. 2003, 5, 3579. DOI: 10.1021/ol035227o
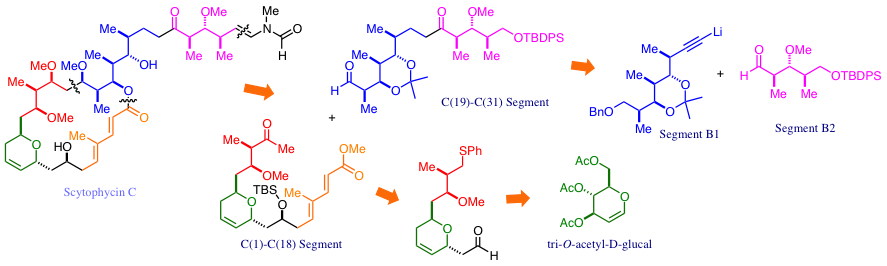
The C2 Selective Nucleophilic Substitution Reactions of 2,3-Epoxy Alcohols Mediated by Trialkyl Borates: The First endo-Mode Epoxide-Opening Reaction through an Intramolecular Metal Chelate.
Sasaki, M.; Tanino, K.; Hirai, A.; Miyashita, M.
Org. Lett. 2003, 5, 1789. DOI: 10.1021/ol034455f

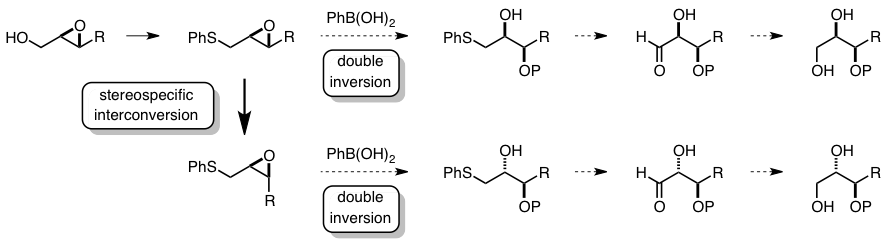
Stereospecific Synthesis of Aldoses Based on the Epoxide-Opening Reaction with Double Inversion of the Configuration.
Hirai, A.; Tonooka, T.; Tanino, K.; Miyashita, M.
Chirality 2003, 15, 108. DOI: 10.1002/chir.10170
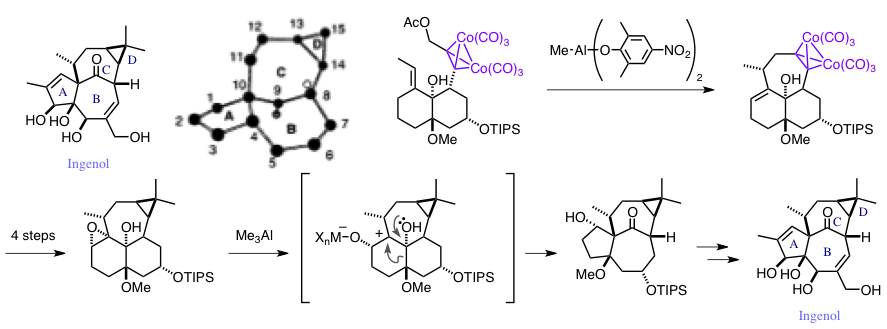
Total Synthesis of Ingenol.
Tanino, K.; Onuki, K.; Asano, K.; Miyashita, M.; Nakamura, T.; Takahashi, Y.; Kuwajima, I.
J. Am. Chem. Soc. 2003, 125, 1498. DOI: 10.1021/ja029226n
 北海道大学 大学院総合化学院 総合化学専攻
北海道大学 大学院総合化学院 総合化学専攻